#apaperaday: Human iPSC model reveals a central role for NOX4 and oxidative stress in Duchenne cardiomyopathy
In today’s #apaperaday, Prof. Aartsma-Rus reads and comments on the paper titled: Human iPSC model reveals a central role for NOX4 and oxidative stress in Duchenne cardiomyopathy.
Today’s pick is from Stem Cell Reports by Duelen et al on cardiac differentiated IPSCs of Duchenne and controls to study oxidative stress and response to dystrophin and antioxidant treatment doi 10.1016/j.stemcr.2021.12.019.
Duchenne is caused by lack of dystrophin, leading to skeletal muscle & heart pathology amongst others. The pathomechanism in skeletal muscle is rather well studied due to the availability of muscle biopsies, but cardiac pathology is studied less well due to lack of heart biopsies.
From mouse studies it is known that calcium homeostasis is disrupted in heart, and there is inflammation, oxidative stress and ischemia. Induced pluripotent stemcells (IPSCs) differentiated into cardiac lineages allow studying these processes in more detail in human cells.
Authors generated IPSCs for Duchenne patients & confirmed pluripotency & lack of dystrophin in cardiac differentiated cells. They generated an isogenic control by correcting a nonsense mutation in exon 35 in the Duchenne IPSCs and confirmed pluripotency and dystrophin production.
Authors analyze the IPCS induced cardiomyocytes and show that the DMD cells have changed electrophysiological parameters, increased cell death due to stressors and higher production of reactive oxygen species (ROS) (i.e. more oxidative stress).
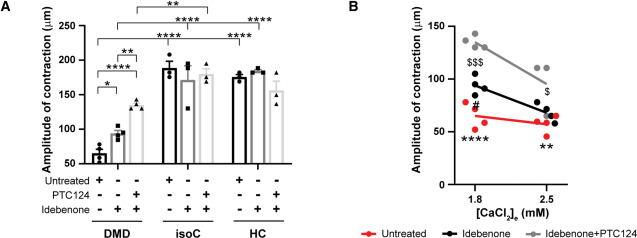
Treatment with PTC124 (restoring dystrophin for nonsense mutation) or idebenone (anti-oxidant & electron shuttle activator) improved pathological findings. I do not like the way authors presented results in panel A makes it hard to see differences between untreated and treated.

Authors show that the DMD IPSC cardiomyocytes have more depolarized mitochondria. Also here treatment with PTC124 and idebenone improved matters. They found overexpression of NOX4 in DMD IPSC cardiomyocytes – also this restored with PTC124 and idebenone.
Authors go into the mechanism through which idebenone improves mitochondria depolarization: NOX4 can bind ATP, which regulates its activity. Idebenone increases ATP availability which reduces ROS production.
Finally, authors show reduced strength for DMD IPSC cardiomyocytes. This increases with idebenone treatment & more with PTC124+idebenone treatment (sadly no PTC124 treatment alone was tested so it is unsure whether it is an additive or synergistic effect or only due to PTC124)
I like that the authors have done a lot of experiments to study pathology and treatment responses. I think however that the title is not well chosen (unduly focusing on NOX4 – while the paper contains a lot more information and findings).
PTC124 is also known as ataluren and this is conditionally approved in Europe for Duchenne patients with nonsense mutations. Idebenone was tested in clinical trials for Duchenne but development was stopped due to lack of therapeutic effects found in interim analysis.
I cannot find anywhere in the paper or the supplementary methods which dose of idebenone was used here (apologies if I missed it..). That makes it difficult to put the results in perspective – was it a dose that was also achieved in clinical trials? Or was it a much higher dose?
After looking for the dose used and not easily finding it, I intend to provide doses of compounds used in figures or figure legends of my own future papers to make it easier for the reader. If someone can enlighten me about the idebenone dose used, that would be much appreciated.
Pictures by Annemieke, used with permission.






