#apaperaday: Evaluation of the dystrophin carboxy-terminal domain for micro-dystrophin gene therapy in cardiac and skeletal muscles in the DMD mdx rat model
In today’s #apaperaday, Prof. Aartsma-Rus reads and comments on the paper titled: Evaluation of the dystrophin carboxy-terminal domain for micro-dystrophin gene therapy in cardiac and skeletal muscles in the DMD mdx rat model.
Today’s pick is from Gene Therapy by Bourdon et al on the evaluation of the additional benefit of adding the C-terminal part of dystrophin to micro-dystrophin in the DMD rat model doi 10.1038/s41434-022-00317-6.
Duchenne is caused by lack of dystrophin. Without dystrophin the dystrophin associated glycoprotein complex (DAPC) cannot form in skeletal muscle and heart, resulting in pathology and eventually replacement of muscle by fibrosis and adipose tissues.
Gene replacement therapy is being developed for Duchenne. However, dystrophin is a very long protein and the genetic code does not fit into the only viral vector that can infect skeletal muscle and heart efficiently in humans (AAV). Thus micro-dystrophins were generated.
Full length dystrophin contains actin binding domains, a central repeat domain with 24 spectrin repeats and 4 hinges, a cysteine rich domain and a c-terminal domain. Micro-dystrophins contain the actin binding domain, 4-5 spectrin repeats, 2-3 hinges and the cystein rich domain.
They all lack the C-terminal domain, even though this is ~36 kDa (~8.5% of the full-length protein) and even though the C-terminal domain does bind to several proteins (e.g. the syntrophins and alpha dystrobrevin) and is known to strengthen the DAPC.
We know micro-dystophin is partially functional in mdx mouse – but this model has a mild phenotype. Here authors used micro-dystrophin in the more severely affected rat mdx model (my abbreviation rdx). They also tested micro-dystrophins with part or full C-terminal domains

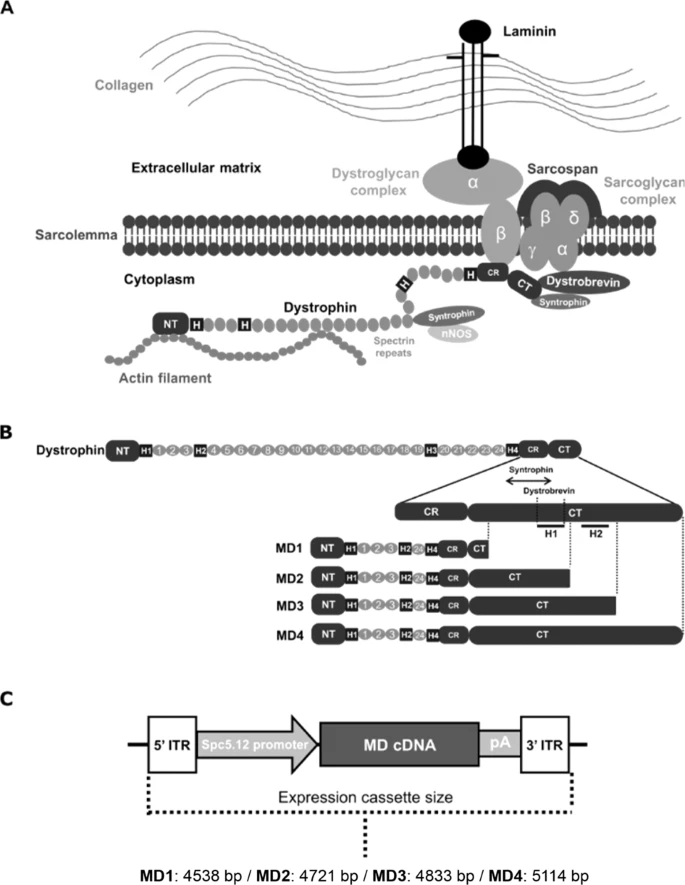
First they tested whether these micro-dystrophin with partial or full C-terminal domains (C+) were properly expressed. 3 were, but the 1 containing the full C-terminal domain was poorly expressed, likely because the size of the micro-dystrophin C+ was too long for AAV.
Authors assessed which proteins the micro-dystrophin & micro-dystrophin C+ constructs could bind using mass-spectrometry in rdx muscle. This revealed the C+ constructs were better binders to many DAPC members than regular micro-dystrophin, sometimes better than normal dystrophin.
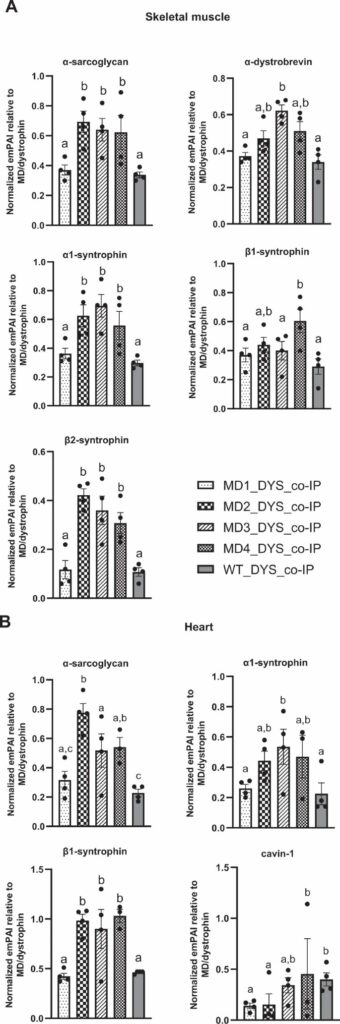
They also studied effect on muscle and heart pathology (histology, calcium homeostasis) and function (strength, eccentric exercise force drop) showing the micro-dystrophin and C+ dystrophins did equally well: better than untreated but worse than wild type rats.
Authors conclude the C+ dystrophins are better binders of DAPC components, but do not improve functional & pathological beneficial effects compared to micro-dystrophin without C-terminal domain IN RATS (authors point this out). Question is of course if this translates to humans.
There are several aspects I like about this work: 1. Authors used proper controls (untreated & wild type rats). 2. Authors used suboptimal dose of AAV to better assess potential benefit of C+ constructs (but did also include the optimal dose for micro-dystrophin as control).
Authors speculate in the discussion that the suboptimal dose might be enough for heart and suggest future studies with a lower dose to study heart effects in more depth. Looking forward to the results (DAPC composition in heart is different from muscle, so interesting question).
Authors also discuss the strange finding that some C+ micro-dystrophins were better DAPC member binders than normal dystrophin. I agree with authors that context matters, and the micro-dystrophins C+ will have structural differences which may explain this.
Finally, the question is if C+ micro-dystrophins would be better than regular micro-dystrophins in humans. This we do not know yet – C+ micro-dystrophin is not tested in clinical trials. For micro-dystrophin, placebo controlled data on functional effects are not yet available.
So for now we do not even know if micro-dystrophin is functional in Duchenne patients. Hopefully the placebo-controlled trials will show this to be the case- and then the C+ micro-dystrophin becomes either more or less relevant depending on how well micro-dystrophin works.
Pictures by Annemieke, used with permission.






