#apaperaday: Engineered skeletal muscle recapitulates human muscle development, regeneration and dystrophy
In today’s #apaperaday, Prof. Aartsma-Rus reads and comments on the paper titled: Engineered skeletal muscle recapitulates human muscle development, regeneration and dystrophy
Today’s pick is from Journal f Cachexia, Sarcopenia and Muscle by Shahriyari et al on an engineered skeletal muscle model (differentiated from IPSCs). Doi 10.1002/jcsm.13094
Studying muscle can be done with 2D cultures with primary myoblasts or immortalized myoblasts. However, these are not fully recapitulating the 3D organization of a muscle fiber. Authors here present a method to differentiate IPSCs into muscle in a transgene and serum free way.
Authors first differentiate cells into mesoderm (the germlayer giving rise to muscle) and then steer towards myogenic lineages. They can use the system to generate 2D and 3D muscle cells, and need a collagen/matrixgel hydrogels. In 3D fibers are grown between metal holders.

This then allows electric stimulation and measuring force when the muscle bundles contract (which they do!) Authors used single cell sequencing, showing their muscles are a mix of muscle, neuronal and mesenchymal cells. They also showed that the fibers produced satellite cells.
Using T3 hormone maturation of the muscles could be improved (later stage myosins). These satellite cells were located on top of the fibers and could be activated with a cardiotoxin injury protocol, and then repaired the damage. I have to say that that is quite cool.
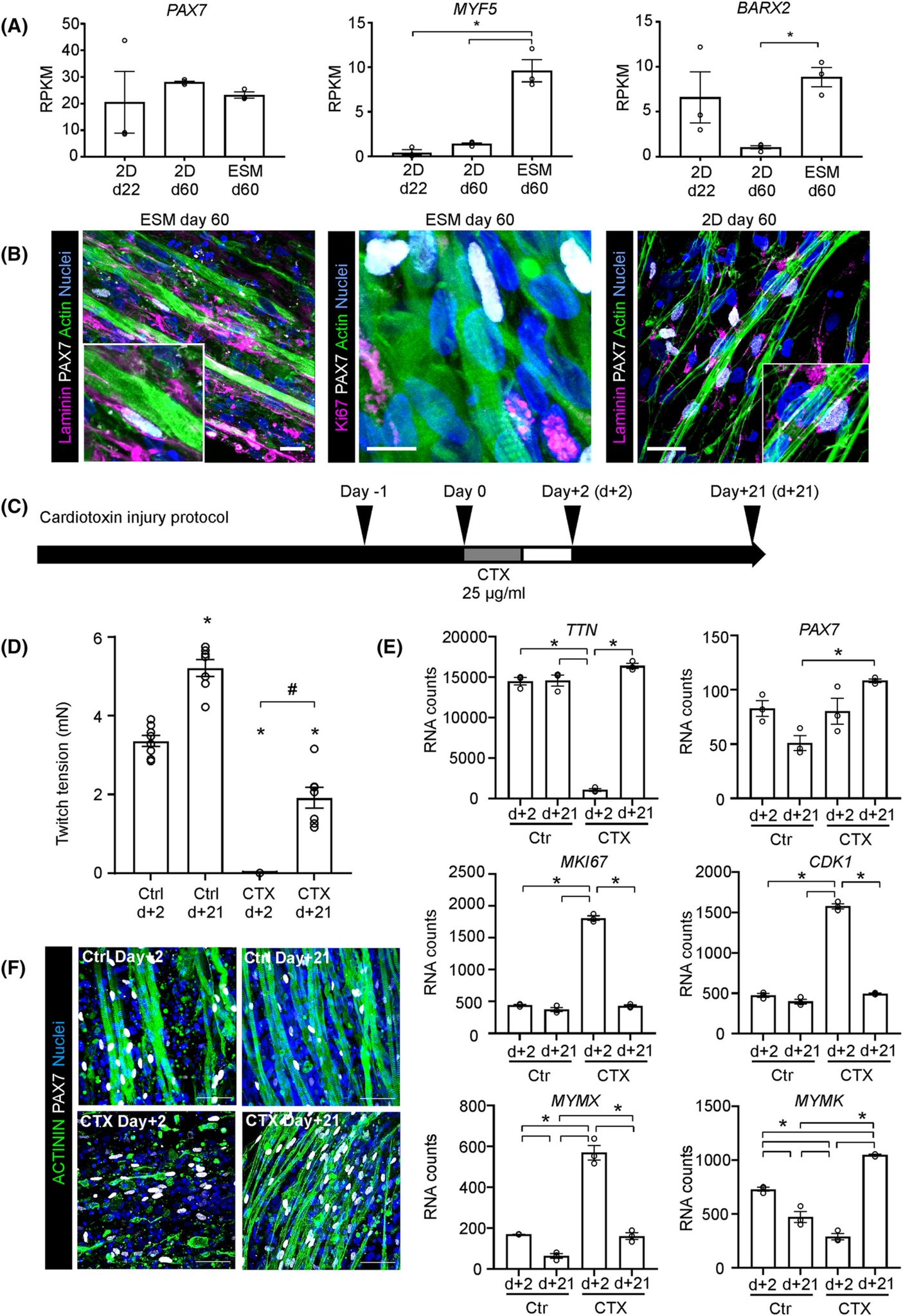
Finally authors tested the model for Duchenne IPSC cells (deletion of exon 48-50) showing that lack of dystrophin did not affect maturation of the cells, but that the muscles without dystrophin had less fast twitch fiber myosin heavy chains.
Authors show a lot of data in the paper, and discuss that having a 3D system is key to producing these ‘engineered skeletal muscles’ and to study muscle force and composition. The fact that they see fast and slow twitch fibers and satellite cells is very nice.
Authors do stress in the discussion that this is a model system for fetal muscle and not for adult muscle – so it is not a perfect model. However, when you are aware of the limitation and you have a research question for which this does not matter, this is a nice additional tool.






