#apaperaday: Dystrophin deficiency affects human astrocyte properties and response to damage
In today’s #apaperaday, Prof. Aartsma-Rus reads and comments on the paper titled: dystrophin deficiency affects human astrocyte properties and response to damage.
Today’s pick is by Lange et al from Glia. It is a study on astrocyte properties in cells with and without dystrophin. The paper went through revisions for 2 years (submitted nov 2019, accepted oct 2021!). Great perseverance from authors! DOI 10.1002/glia24116
dystrophin has multiple isoforms. The full-length one (Dp427) is expressed in cardiac and skeletal muscle and in the central nervous system. Multiple other isoforms (Dp71, Dp140, Dp40 and their alternatively spliced forms) are expressed primarily in the central nervous system.
The role of dystrophin in the brain has not been well studied. It is known that in mdx mice a reduction in GABAergic synapses results in increased anxiety and fear. In this work authors focused on astrocytes (the support system of the CNS and part of the blood brain barrier).
We already showed lack of dystrophin results in astrocyte dysfunction in a collaborative effort with Samie Patel https://pubmed.ncbi.nlm.nih.gov/31434868/ Here authors confirm our findings and extend on the work (science in action :))
First authors studied expression dystrophin in embryonic human brain lysates. This showed some expression of Dp427, no Dp260 and high expression of Dp140, Dp71 and Dp40. Results were confirmed with 2 & different antibodies (which were specific – confirmed in DMD lysate).
Then authors differentiated 2 control and 3 DMD (2 lacking all isoforms, 1 lacking Dp71 and Dp116) IPSCs into neuronal progenitor cells and astrocytes. This worked for DMD and control cells. However, aquaporin 4 expression was higher in DMD cells (see figure).
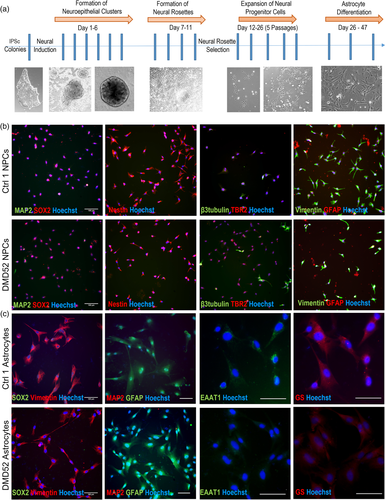
DMD astrocytes proliferated more and had higher metabolic activity. Also, they were more sensitive to oxidative stress, while they did not respond to inflammatory stressors while control cells did (likely because they were already in a stressed condition)
RNAseq analysis revealed lower expression of genes involved in neuronal development, increased inflammatory pathways, increased metabolic activity and dysregulation of calcium for DMD astrocytes.
Note that 2 of the 3 DMD cell lines used lacked all dystrophin isoforms. This is very rare. Authors outline that the results seen for the astrocytes lacking only Dp427, Dp260 and Dp140 showed similar results, suggesting these are generalizable results.
However, I agree with authors that more work is needed. E.g. 50% of Duchenne patients can produce Dp140. Currently dystrophin function in human brains (post mortem), Duchenne and Becker patients (imaging) and animal models is studied in more detail in BIND.
Authors also discuss dystrophin forms likely have a function in development of the brain but also in the maintenance. This has consequences for therapeutic interventions (restoring dystrophin in the brain will not undo developmental errors, but may improve ‘maintenance’ functions). This is also something that is studied further in BIND. Stay tuned for updates!
Pictures by Annemieke, used with permission.






