#apaperaday: Paeonia lactiflora extract improves the muscle function of mdx mice
In today’s #apaperaday, Prof. Aartsma-Rus reads and comments on the paper titled: Paeonia lactiflora extract improves the muscle function of mdx mice, an animal model of Duchenne muscular dystrophy, via downregulating the high mobility group box 1 protein.
Today’s pick is by Sim et al on the anti-inflammatory properties of paeonia lactiflora in the mdx mouse model from the journal of ethnopharmacology (the study of herbs etc for medicinal use by ethnic groups). Doi 10.1016/j.jep.2022.115079
Authors outline Duchenne has a strong inflammatory component. At the same time, Paeonia lactiflora (PL) is a member of the peony family used in Asia for its anti-inflammatory properties. Here authors test whether PL can reduce inflammation in mdx mouse and improve pathology
Authors us male mdx mouse, a wild type reference and a prednisone comparator (triple hooray for good study set up!) Animals are treated for 4 weeks starting at 5 weeks of age and then analyzed. PL treatment increased grip strength and rotarod running in a dose dependent manner.
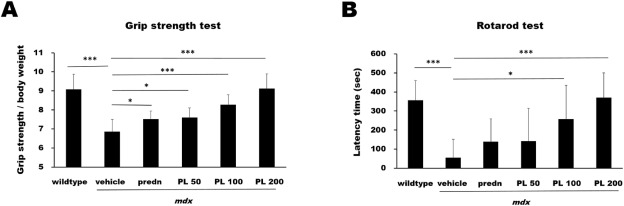
What is strange is that authors do not mention how PL compares to the prednisone comparator in the abstract or the result – it is only shown in the figure. Also missing are the weights of the mice – prednisone is expected to reduce weight – what about PL?
Absolute force is missing as well – if there is significant weight loss (as is expected with prednisone treatment) the force corrected for body weight can increase while absolute force is unchanged. Important to show both in case bodyweight changes. Still not sure if PL changes this.
Authors also look at histology with H&E. They show 4 images of gastrocnemius that are zoomed in. This shows less degeneration in prednisone treated mice and further reduction in PL treated mice (dose dependent). However, no quantification of whole cross section was done.
Also I am disappointed the authors only looked at the gastrocnemius and not also at the much more severely affected diaphragm. This is much more relevant than a slight reduction in the hardly affected gastrocnemius muscle.
Serum creatine kinase is reduced a little bit in prednisone and PL treated mice. Authors then perform detailed proteomic analysis in mdx (+ and – PL treatment) and wild type. The prednisone treated group is used in the validation studies.
The proteomic study shows that in mdx there is more protein production than wild type (likely reflecting active regeneration). In PL treated mdx mice, inflammatory proteins are reduced and there is less oxidative stress. In serum NFKB (inflammation master regulator) is reduced.
I commend the authors for the detailed analysis and the validation studies – this underlines the anti-inflammatory properties of the PL extract. However, again authors use the gastrocnemius. The diaphragm would have been so much more informative due to severity.
Authors discuss that prednisone is currently the standard of care but due to the side effects, alternatives are needed. PL could be an alternative – I like that authors are very cautious and do not extrapolate immediately to patients. Instead they ask the right questions.
PL is tolerated at much higher levels than used in the mdx mice here. However, there is as yet little data on chronic use. More studies are needed but authors point out the mechanism of action of PL is now confirmed (anti-inflammatory).
Before thinking of planning clinical trials I think more studies are needed to see what the effects are on the diaphragm of mdx mouse and/or whether PL treatment can also reduce the more severe pathology in e.g. the d2/mdx mouse or the rat mdx model.
Also it is not yet clear which dose is optimal. For some analyses the highest dose performed best, for others maximum treatment effect was reached with a lower dose – but that was all in the mildly affected gastrocnemius. Dose optimization needs diaphragm analysis.
In summary, authors did many things right and took along all control groups that were needed. They also confirmed the anti-inflammatory effect of PL. However, they should have also analyzed the diaphragm.
Furthermore, they should have mentioned in the discussion that as another alternative to prednisone vamorolone is currently tested in clinical trials for Duchenne.
Pictures by Annemieke, used with permission.






